Uni Cei En Iso 14971 Risk
Panzer General 3d Deutsch here. Url Helper 3 45 Serial Killer on this page. General • U.S. Regulations, 21 CFR Part 820 for a Class II medical device • Directive 93/42/CEE annex II • UNI CEI EN ISO:“ Medical devices -- Quality management systems -- Requirements for regulatory purposes” • UNI CEI EN ISO 14971:2012: Medical devices -- Application of risk management to medical devices • CEI EN: Medical device software - Software life-cycle processes • UNI EN ISO 9004:2009 Managing for the sustained success of an organization -- A quality management approach. • UNI EN ISO 9000:2005 Quality management systems -- Fundamentals and vocabulary • CEI EN: Medical electrical equipment. Recurrent test and test after repair of medical electrical equipment • CEI UNI EN ISO 15223-1:2012 Medical devices -- Symbols to be used with medical device labels, labelling and information to be supplied -- Part 1: General requirements • IEC “Medical devices – Application of usability engineering to medical devices”.
Safety • IEC 60601-1:2005: Medical electrical equipment – Part 1: General requirements for basic safety and essential performance • IEC 60601-1-6:2010: Medical electrical equipment -- Part 1-6: General requirements for basic safety and essential performance - Collateral standard: Usability • IEC 60601-1-11:2010: Medical electrical equipment -- Part 1-11: General requirements for basic safety and essential performance - Collateral standard: Requirements for medical electrical equipment and medical electrical systems used in the home healthcare environment. Free Clone Craigslist Programming.
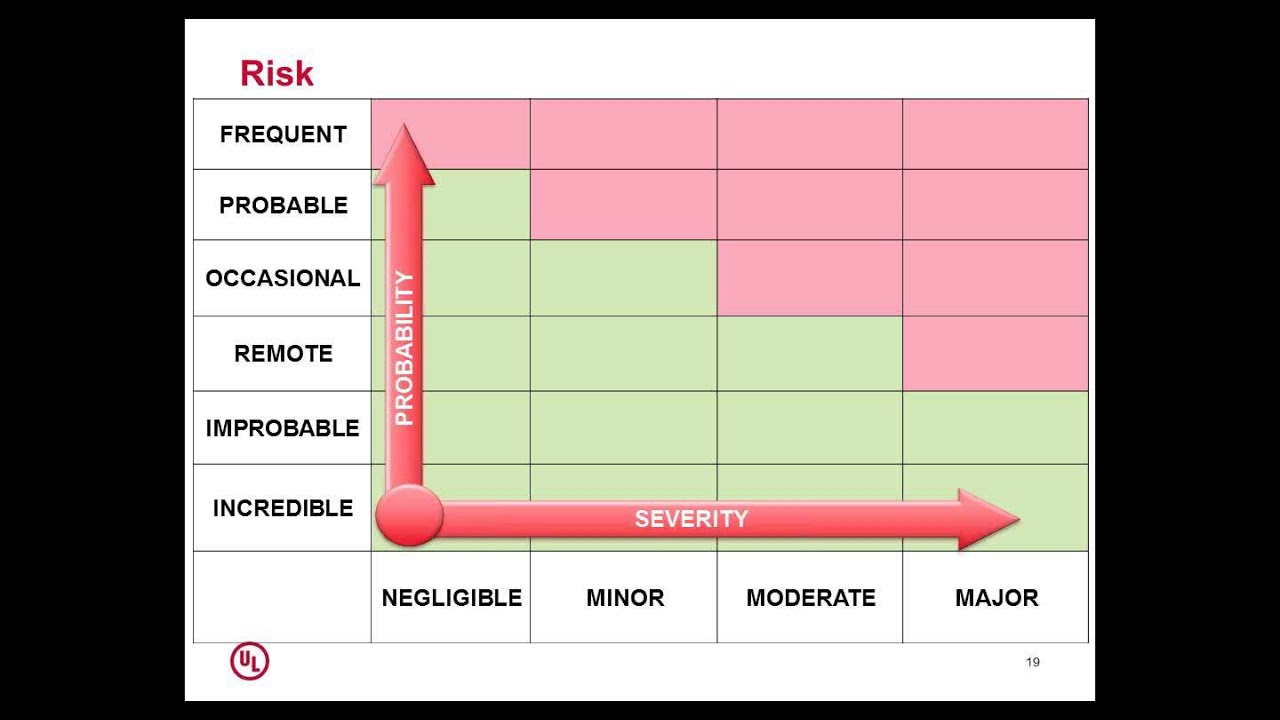
The purpose of ISO 14971 is to help manufacturers to establish a medical device risk management process that can be used to identify hazards, to estimate and evaluate. Published in English, French and Spanish, six times per year, ISOfocus is your gateway to International Standards. Whether a multinational enterprise. ISO 14971: Medical devices - Application of risk management to medical devices. ISO 14971 2nd Edition. BS EN 12442-1 November 15, 2000.
ISO 14971 is an standard for the application of risk management to. The ISO Technical Committee responsible for the maintenance of this standard is ISO TC 210 working with IEC/SC62A through Joint Working Group one (JWG1). This standard is the culmination of the work starting in, and. The latest significant revision was published in 2007 with a minor update published in 2009. In 2013, a technical report ISO/TR 24971 was published by ISO TC 210 to provide expert guidance on the application of this standard. This standard establishes the requirements for risk management to determine the safety of a medical device by the manufacturer during the product life cycle. Such activity is required by higher level regulation and other quality management system standards such as.